
Joseph Feher, in, 2017 Endogenous Creatinine Clearance Approximates the GFRCreatinine is a by-product of muscle metabolism in which creatine in the muscle is converted nonenzymatically to creatinine. Because the total body content of creatine is fairly constant, there is a continual production of creatinine and a continual excretion of it in the urine. The typical 70-kg adult man produces about 2 g of creatinine per day.
Creatinine is formed as a result of the natural breakdown of your muscle tissue and can then enter the blood. Under normal conditions, your kidneys filter creatinine from your blood. Creatinine leaves the body through urine. What is the creatinine clearance test? The creatinine clearance is a test that allows your doctor to assess your kidney.
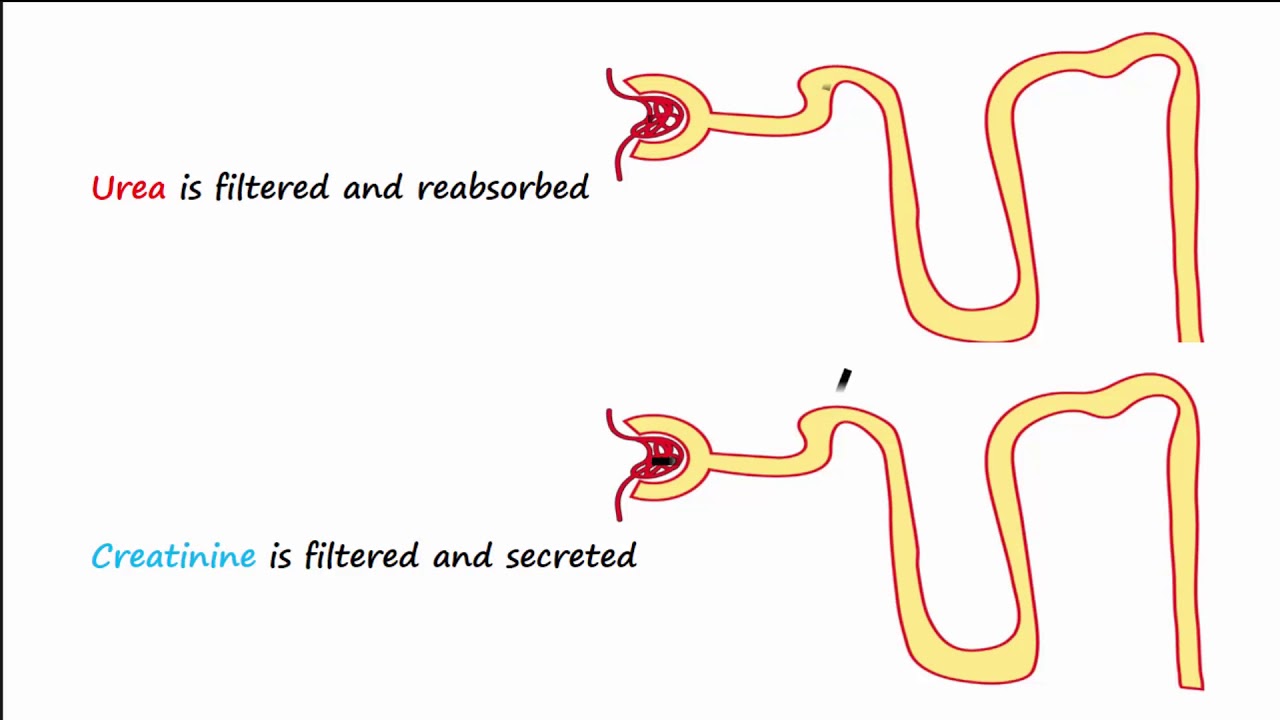
As evident from Figure 7.4.5, creatinine is slightly secreted by the kidneys so that at low plasma creatinine the clearance of creatinine is about 5–10% greater than the inulin clearance. However, creatinine is already present in the blood at steady-state levels, so the endogenous creatinine clearance can be used without the necessity of having to infuse inulin. All that is necessary is a timed urine sample and a plasma sample. Measuring the creatinine in both the urine and plasma sample allows the calculation of the endogenous creatinine clearance that provides an approximate measure of the GFR.
In, 2011 Creatinine ClearanceMeasuring the creatinine clearance rate obviates some of the problems of using serum creatinine concentration as a marker of GFR. Creatinine clearance should not be affected by differences in steady-state creatinine production due to differences in muscle mass that affect serum creatinine level. However, the reliability of creatinine clearance is greatly diminished by variability in tubular secretion of creatinine and by the inability of most patients to accurately collect timed urine samples. Prolonged storage of the urine can also introduce error, as high temperature and low urine pH enhance the conversion of creatine to creatinine in stored urine. Therefore, urine samples should be refrigerated and the urine creatinine level measured without undue delay. Creatinine is generated from the breakdown of creatine in muscle, distributed throughout total body water, and excreted by the kidneys primarily by glomerular filtration.
Although the serum level is affected primarily by the level of GFR, it is also affected by other physiologic processes, such as tubular secretion, generation, and extrarenal excretion of creatinine. Table 3.2 lists the factors that limit use of creatinine as estimated GFR.
Due to variation in these processes among individuals and over time within individuals, especially creatinine generation, the cutoff for normal versus abnormal serum creatinine concentration differs among groups. In addition, assays for serum creatinine vary across clinical laboratories, leading to differences in GFR estimates for the same patient when creatinine is measured in different labs. Levey, in, 2010 Creatinine Metabolism and ExcretionCreatinine is a 113-d end product of muscle catabolism. 1 Advantages of creatinine include its ease of measurement and the low cost and widespread availability of assays. Disadvantages include the large number of non-GFR determinants (see Fig. 3.2), leading to a wide range of GFR for a given plasma creatinine level.
For example, a serum creatinine level of 1.5 mg/dl (132 µmol/l) may correspond to a GFR from approximately 20 to 90 ml/min per 1.73 m 2. Factors affecting serum creatinine concentration. (Reprinted with permission from reference 5.)Creatinine is released into the circulation at a constant rate. It is not protein bound and is freely filtered across the glomerulus and secreted by the tubules. Several medications, such as cimetidine and trimethoprim, competitively inhibit creatinine secretion and reduce creatinine clearance.
These medications thus lead to a rise in the serum creatinine concentration without an effect on GFR (see Fig. 3.4).In addition, creatinine is contained in intestinal secretions and can be degraded by bacteria. If GFR is reduced, the amount of creatinine eliminated through this extrarenal route is increased. Antibiotics can raise serum creatinine concentration by destroying intestinal flora, thereby interfering with extrarenal elimination, as well as by reduction of the GFR.
The rise in serum creatinine concentration after inhibition of tubular secretion and extrarenal elimination is greater in patients with a reduced GFR. Clinically, it can be difficult to distinguish a rise in serum creatinine concentration due to inhibition of creatinine secretion or extrarenal elimination from a decline in GFR, but processes other than a decline in GFR should be suspected if serum urea concentration remains unchanged despite a significant change in serum creatinine concentration in a patient with an initially reduced GFR.Creatinine clearance is usually computed from the creatinine excretion in a 24-hour urine collection and single measurement of serum creatinine in the steady state. In a complete collection, creatinine excretion should be approximately 20 to 25 mg/kg per day and 15 to 20 mg/kg per day in healthy young men and women, respectively, and deviations from these expected values can give some indication of errors in timing or completeness of urine collection.
Creatinine clearance systematically overestimates GFR because of tubular creatinine secretion. In the past, the amount of creatinine excreted by tubular secretion at normal levels of GFR was thought to be relatively small (10% to 15%), but with newer, more accurate assays for low values of serum creatinine, it appears that this difference may be substantially greater. At low values of GFR, the amount of creatinine excreted by tubular secretion may exceed the amount filtered. Jean-Pierre Guignard, in, 2017 CreatinineCreatinine is the anhydride of creatine, a compound that exists in skeletal muscle as creatine phosphate. It has a molecular mass of 113 Da. Conversion of creatine to creatinine is nonenzymatic and irreversible. The serum creatinine level reflects total body supplies of creatine and correlates with muscle mass.
Creatinine is excreted through the kidneys in quantities proportional to the serum content. The renal excretion of endogenous creatinine is very similar to that of inulin in humans and several animal species. However, in addition to being filtered through the glomerulus, creatinine is secreted in part by the renal tubular cells. In spite of this, creatinine clearance correlates well with inulin clearance when the GFR is normal. This agreement results from the balance of two factors: (1) the excretion rate of creatinine is higher than the filtered rate because of the occurrence of tubular secretion of creatinine, and (2) the measured plasma creatinine concentration is higher than the true creatinine concentration because of the presence of non-creatinine chromogens that interfere with the colorimetric analysis of creatinine (Jaffe reaction).Overestimation of GFR by creatinine clearance is usually more evident at low GFR. As GFR falls progressively during the course of renal disease, the renal tubular secretion of creatinine contributes an increasing fraction to urinary excretion, so creatinine clearance may substantially exceed the actual GFR.The use of creatinine clearance to estimate GFR may be poorly reliable in uremic patients. Creatinine is uniformly distributed in the body water, and it diffuses into the gut.
At a normal plasma concentration, the amount of creatinine entering the gut is negligible; it may become significant during renal failure, when the plasma creatinine concentration increases. 23 This phenomenon may also explain why creatinine clearance overestimates true GFR in patients with renal failure.Although in use for decades, the methods available for the chemical determination of creatinine still present important drawbacks. As noted above, the traditional assay for measuring creatinine) (the Jaffe reaction) substantially overestimates true serum creatinine levels because of the presence of interfering pseudochromogenic constituents in the blood. The major drawback for routine use in neonates is interference by bilirubin.
Adaptations of the alkaline picrate assay have reduced the overestimation without totally eliminating the interference. 24 Although more specific than the Jaffe method, the enzymatic techniques are still biased by various interfering substances. 24 A new method coupling high-performance liquid chromatography and isotope dilution mass spectrometry appears to have an excellent specificity and low relative standard deviation. 25 Creatinine as a Marker of Glomerular Filtration Rate in Neonates.
The plasma creatinine concentration varies during the first postnatal weeks. 26,27 It is elevated at birth and decreases rapidly during the first week of life ( Figure 103-3); values stabilize at approximately 0.40 mg/dL (35 µmol/L; range, 0.14 to 0.70 mg/dL 12 to 61 µmol/L) on the fifth postnatal day in term infants and somewhat later in very-low-birth-weight infants. 27,28 The elevated plasma creatinine concentration at the time of birth reflects maternal creatinine levels. Indeed, a perfect equilibrium between fetal and maternal plasma creatinine concentrations has been observed throughout gestation.
29,30 In very premature neonates, the elevated plasma creatinine concentration at birth increases transiently; the highest levels are reached by the third day of life ( Figure 103-4 and Table 103-1). 31,32 The plasma urea level also rises significantly over time, but it does so in a more variable manner. This transient postnatal increase in plasma creatinine concentration is probably the consequence of creatinine reabsorption (back diffusion) across leaky tubules, 33 as suggested by studies in piglets and newborn rabbits. 33,34 Plasma creatinine levels may take a month to reach neonatal levels in very-low-birth-weight infants.
15) Ucr × V = 21. 115 × age ( women )where creatinine excretion is expressed in mg/kg/d and age is expressed in years.These equations do not take into account racial and ethnic differences in muscle mass. African American (black) males and females have higher muscle mass and consequently higher creatinine excretion than their Caucasian (white) counterparts. Asians have lower muscle mass and lower creatinine excretion rate than their Caucasian counterparts. 49,50 Other equations to estimate creatinine excretion have been proposed, but precision is limited; approximately 20% of estimates deviate by more than 30% of measured creatinine excretion rate. 51Creatinine generation is affected by diet and disorders of skeletal muscle. Muscle wasting is associated with a decreased creatine pool, leading to decreased creatinine generation and excretion.
Reduction in dietary protein causes a decrease in the creatine pool by 5% to 15%, probably by reducing the availability of creatine precursors. Of greater importance is the effect of creatine in the diet. Creatine is contained largely in meat; elimination of creatine from the diet decreases urinary creatinine excretion by as much as 30%. Conversely, ingesting a creatine supplement increases the size of the creatine pool and increases creatinine excretion. Meat intake also affects creatinine generation and excretion independent of its effect on the creatine pool. During cooking, a variable amount of the creatine in meat is converted to creatinine, which is absorbed from the gastrointestinal tract. After ingestion of cooked meat, there is a sudden transient increase in the serum creatinine concentration and urinary creatinine excretion.
Creatinine is a spontaneously formed cyclic degradation production of creatine breakdown in the tissues. Creatine is synthesized in the liver, and to a lesser extent the kidney, and acquired in a meat-rich diet. It is taken up into cells via transmembrane transporters, whereas phosphocreatine, it provides a buffer for rapid energy production. Skeletal muscle thus contains most of the body's pool of creatine and consequently is the source the majority of creatinine production.
3 As a small, 113 Da molecule, creatinine is filtered freely in the glomerulus and appears unaltered in the urine together with a small amount of active tubular secretion, whereas extrarenal creatinine excretion is negligible in most circumstances. Thus as plasma creatinine is excreted predominantly by glomerular ultrafiltration: when the GFR decreases, creatinine accumulates. However, to understand the meaning of baseline plasma creatinine and acute changes in its concentration, an understanding of the steady state and dynamic kinetics of creatinine generation and excretion is required. First, because all creatinine freely filtered in the glomerulus is excreted in the urine, and this constitutes more than 90% of creatinine excretion, net creatinine excretion approximates to the GFR (rate of plasma filtration) multiplied by the concentration of creatinine in the plasma. Second, at steady state (defined by constant plasma creatinine), rate of excretion must equal rate of creatinine generation in the following relationship. Thus, when at steady state, GFR is proportional to the reciprocal of the plasma creatinine ( Fig.
With the assumption that a creatinine change occurs and stabilizes at the new level, achieving a new steady state, a doubling of plasma creatinine therefore implies an approximate halving of GFR, as long as the creatinine generation rate also remains constant. This relationship forms the basis of the use of 1.5-, two-, and threefold rises in creatinine above a baseline value to define severity of AKI in consensus definitions, reflecting proportionate decrease in underlying GFR. However, although relative changes in plasma creatinine define AKI, there are significant limitations to the acute interpretation of these values in the critically ill. 4,5 First, use of plasma creatinine as a reflection of the change in GFR depends on a new steady-state creatinine concentration. After a change in GFR, creatinine will rise or fall until a new steady state is achieved, where creatinine excretion at a high plasma creatinine concentration again matches production, the duration of this process being dependent on the magnitude of change in GFR and the underlying creatinine generation rate. With slow changes in GFR, such as progressive chronic kidney disease, creatinine rises will keep up with falling GFR, and the relationship will remain accurate.
However, in AKI, after a large acute fall in GFR, many days could be required for a new steady state to be achieved and, until then, plasma creatinine will underestimate severity of renal dysfunction. This implies that diagnosis of AKI is likely to be delayed. Second, changes in creatinine generation will alter measured plasma creatinine concentration as much as change in excretion. Creatinine production will fall if there is a reduction in muscle mass, if there is a reduction in the dietary intake of creatine, and in the presence of liver disease. Large and sustained reductions in muscle mass have been observed in the critically ill; similarly a meat-free diet is almost universal in the intensive care unit (ICU). Thus large reductions in creatinine generation may be observed in critical illness, 6 which, in the absence of AKI, are in turn associated with marked reduction in creatinine levels below baseline that persist to hospital discharge.
7 Thus incidence and severity of creatinine-assessed AKI may be underestimated in the critically ill, and, similarly, renal recovery after AKI may be overestimated significantly. 8 Because reduction in creatinine generation is related to duration of critical illness and risk of death, this confounding effect on AKI diagnosis is more significant in sicker patients. These effects are illustrated in Fig. More delayed AKI diagnosis in older, sicker patients for the same GFR reduction is a major confounder when using AKI criteria to define early versus late clinical interventions for AKI in the ICU.
Kinetic modeling 9 of changes in plasma creatinine concentration after an acute 90% reduction in glomerular filtration rate (GFR) after major surgery in a 65-year-old male with a baseline creatinine of 80 µmol/L. While with such severe reduction in GFR AKI stage I criteria is achieved rapidly on a 26.5 µmol/L increase; full severity of AKI would take more than 15 hours to be apparent. Moreover, when a clinician factors in a creatinine generation rate (G) decreasing to 60% of baseline over 48 hours and then the effect of a continuous positive fluid balance of 150 mL/hr over the same period, creatinine increase is retarded significantly, delaying AKI stage II diagnosis by more than 6 hours and AKI stage III diagnosis by up to 1 day, despite likely greater illness severity.

